Monitoring Report Template Clinical Trials

Is investigator properly supervising other personnel.
Monitoring report template clinical trials. The nccih clinical research toolbox provides a web based information repository for investigators and staff involved in nccih funded clinical research. Please note that this page has been updated for 2015 following a quality check and review of the templates and many new ones have been added. Documented in the monitoring visit report and the plan amended if. Completed by the clinical site monitor to document dates and purpose of clinical site monitoring visits.
14 do patient notes have the clinical trial sticker inside the front cover. The sponsor risk assessment form and the trial risk based monitoring strategy appendices 2 3 to sop s 1007 will facilitate the development of the monitoring plan. The toolbox contains templates sample forms and information materials to assist clinical investigators in the development and conduct of high quality clinical research studies. Clinical monitoring plan template.
Phs conversion request memo. Ccr standard operating procedure sop template. Is investigator devoting enough time for the study. Confidentiality agreement clinical investigation agreement and budget letter of indemnification c.
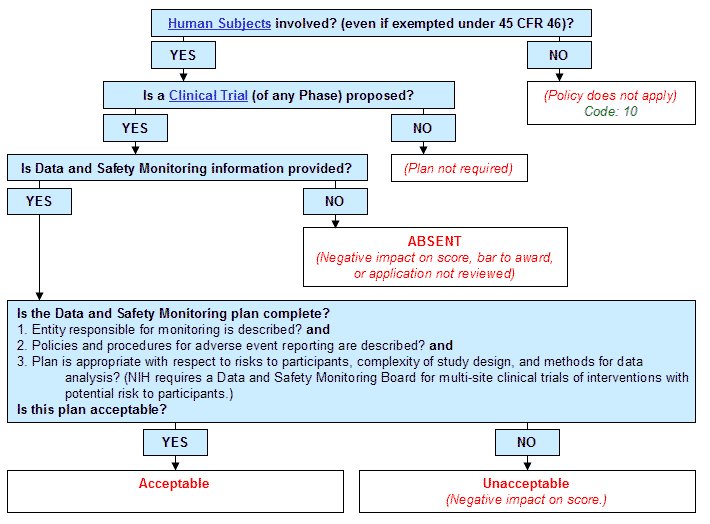
Study monitoring plan template sponsor reference number. Monitoring visit report. A well written monitoring report is an essential part of documenting clinical trial oversight. Preparing to apply for a u01 clinical trial registering with clinicaltrialsgov patient research registries clinical trial policies guidelines and templates.
Workloadfacility status has investigator accepted new studies since last visit. Investigator initiated clinical research data and safety monitoring guidelines and policies clinical study templates and forms nih and other federal guidelinespolicies for clinical research. 15 if yes has this been completed. Welcome to global health trials tools and templates library.
The monitoring risk category. Research team present role rd personnel present role. In addition to being required by iso and ich guidelines it also tells the story of the clinical trial to the fda demonstrating site performance and sponsor oversight during an fda inspection. Ms word template to be used as a starting point for preparing a clinical monitoring plan.
Includes proposed structure draft language and guidance. In the issue that you are making security frameworks for your additional venture or business at that tapering off you could be searching for approaches to set aside times and cash and conclusive your bustle faster. Site monitoring visit report template.